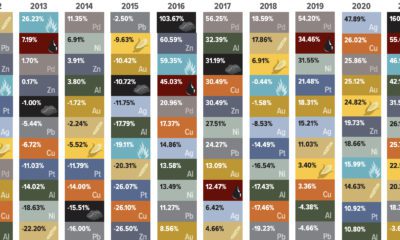
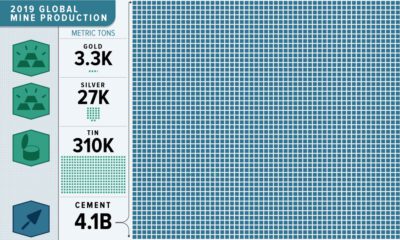
Mining
Aluminum: The Metal Extraordinaire
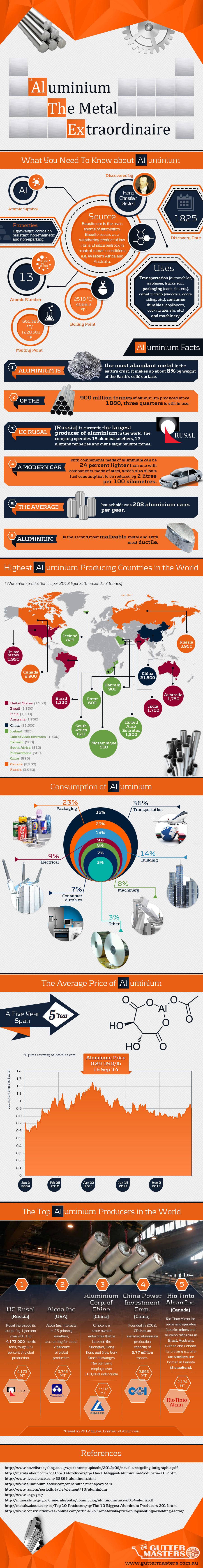
Aluminum: The Metal Extraordinaire
Aluminum (or aluminium) is the world’s most common metal by crustal abundance, making up 8.2% of mass. It’s more common than iron (5.6%) and a whopping 1200x more abundant than copper.
Despite its prevalence, aluminum was not isolated all the way until 1827. This is because it occurs only in compounds, and never in a free form. It also turns out that removing aluminum from these compounds is quite difficult, and an inexpensive process wasn’t discovered until 1886 by a college student in the United States. Charles Martin Hall was interested in the problem, and ran an electric current through a molten mixture of cryolite and aluminum oxide in a wood shed behind his house.
That dropped the price of aluminum drastically, and it became a household metal. Behind iron, aluminum is now the second most used metal in the world. Aluminum can now be found in everything: transportation (planes, cars, and more), buildings, machinery, consumer durables, packaging, and electrical uses.
Original graphic from: GutterMasters
Copper
Brass Rods: The Secure Choice
This graphic shows why brass rods are the secure choice for precision-machined and forged parts.

Brass Rods: The Secure Choice
The unique combination of machinability and recyclability makes brass rods the secure choice for manufacturers seeking future-proof raw material solutions.
This infographic, from the Copper Development Association, shows three ways brass rods give manufacturers greater control and a license to grow in the competitive market for precision-machined and forged products.
Future-Proof Investments in New Machine Tools
A material’s machinability directly impacts machine throughput, which typically has the largest impact on machine shop profitability.
The high-speed machining capabilities of brass rods maximize machine tool performance, allowing manufacturers to run the material faster and longer without sacrificing tool life, chip formation, or surface quality.
The high machining efficiency of brass leads to reduced per-part costs, quicker return on investment (ROI) for new machine tools, and expanded production capacity for new projects.
Supply Security Through Closed Loop Recycling
Brass, like its parent element copper, can be infinitely recycled.
In 2022, brass- and wire-rod mills accounted for the majority of the 830,000 tonnes of copper recycled from scrap in the United States.
Given that scrap ratios for machined parts typically range from 60-70% by weight, producing mills benefit from a secure and steady supply of clean scrap returned directly from customers, which is recycled to create new brass rods.
The high residual value of brass scrap creates a strong recycling incentive. Scrap buy back programs give manufacturers greater control over raw material net costs as scrap value is often factored into supplier purchase agreements.
Next Generation Alloys for a Lead-Free Future
Increasingly stringent global regulations continue to pressure manufacturers to minimize the use of materials containing trace amounts of lead and other harmful impurities.
The latest generation of brass-rod alloys is engineered to meet the most demanding criteria for lead leaching in drinking water and other sensitive applications.
Seven brass-rod alloys passed rigorous testing to become the only ‘Acceptable Materials’ against lower lead leaching criteria recently adopted in the national U.S. drinking water quality standard, NSF 61.

Learn more about the advantages of brass rods solutions.

-

 Base Metals1 year ago
Base Metals1 year agoRanked: The World’s Largest Copper Producers
Many new technologies critical to the energy transition rely on copper. Here are the world’s largest copper producers.
-
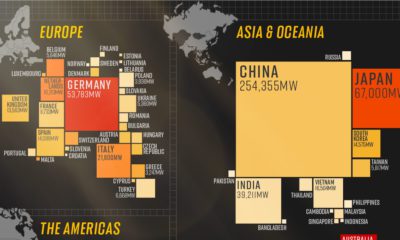
 Silver2 years ago
Silver2 years agoMapped: Solar Power by Country in 2021
In 2020, solar power saw its largest-ever annual capacity expansion at 127 gigawatts. Here’s a snapshot of solar power capacity by country.
-

 Batteries5 years ago
Batteries5 years agoVisualizing Copper’s Role in the Transition to Clean Energy
A clean energy transition is underway as wind, solar, and batteries take center stage. Here’s how copper plays the critical role in these technologies.
-
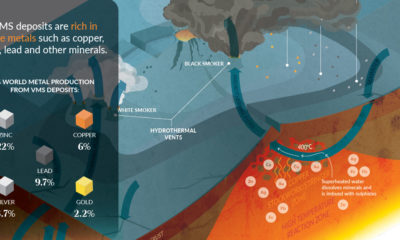
 Science5 years ago
Science5 years agoEverything You Need to Know on VMS Deposits
Deep below the ocean’s waves, VMS deposits spew out massive amounts of minerals like copper, zinc, and gold, making them a key source of the metals…
-

 Copper5 years ago
Copper5 years agoHow Much Copper is in an Electric Vehicle?
Have you ever wondered how much copper is in an electric vehicle? This infographic shows the metal’s properties as well as the quantity of copper used.
-
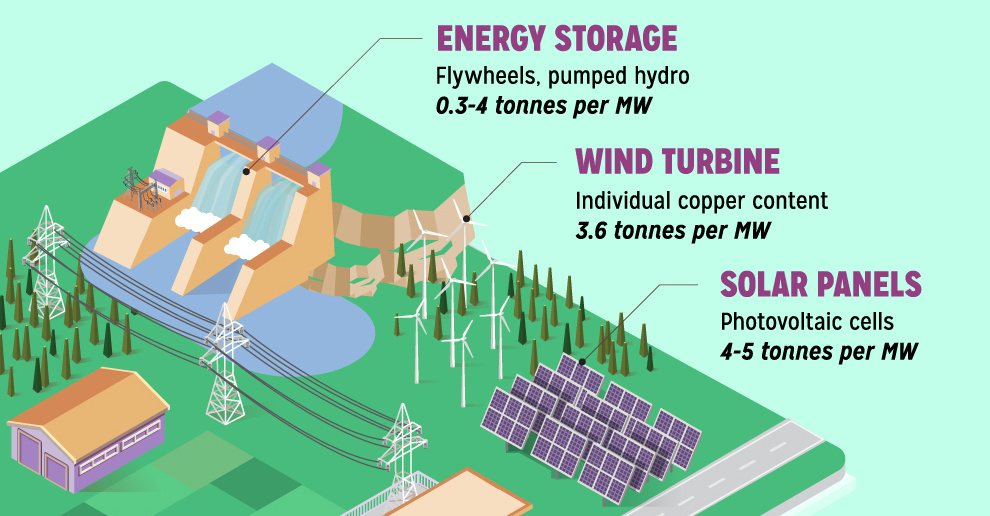
 Copper6 years ago
Copper6 years agoCopper: Driving the Green Energy Revolution
Renewable energy is set to fuel a new era of copper demand – here’s how much copper is used in green applications from EVs to photovoltaics.
-
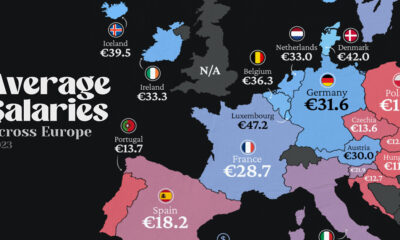
 Maps2 weeks ago
Maps2 weeks agoMapped: Average Wages Across Europe
-

 Money1 week ago
Money1 week agoWhich States Have the Highest Minimum Wage in America?
-

 Real Estate1 week ago
Real Estate1 week agoRanked: The Most Valuable Housing Markets in America
-

 Markets1 week ago
Markets1 week agoCharted: Big Four Market Share by S&P 500 Audits
-
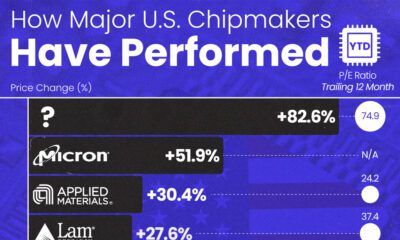
 AI1 week ago
AI1 week agoThe Stock Performance of U.S. Chipmakers So Far in 2024
-
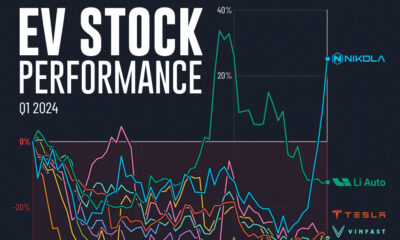
 Automotive1 week ago
Automotive1 week agoAlmost Every EV Stock is Down After Q1 2024
-

 Money2 weeks ago
Money2 weeks agoWhere Does One U.S. Tax Dollar Go?
-

 Green2 weeks ago
Green2 weeks agoRanked: Top Countries by Total Forest Loss Since 2001